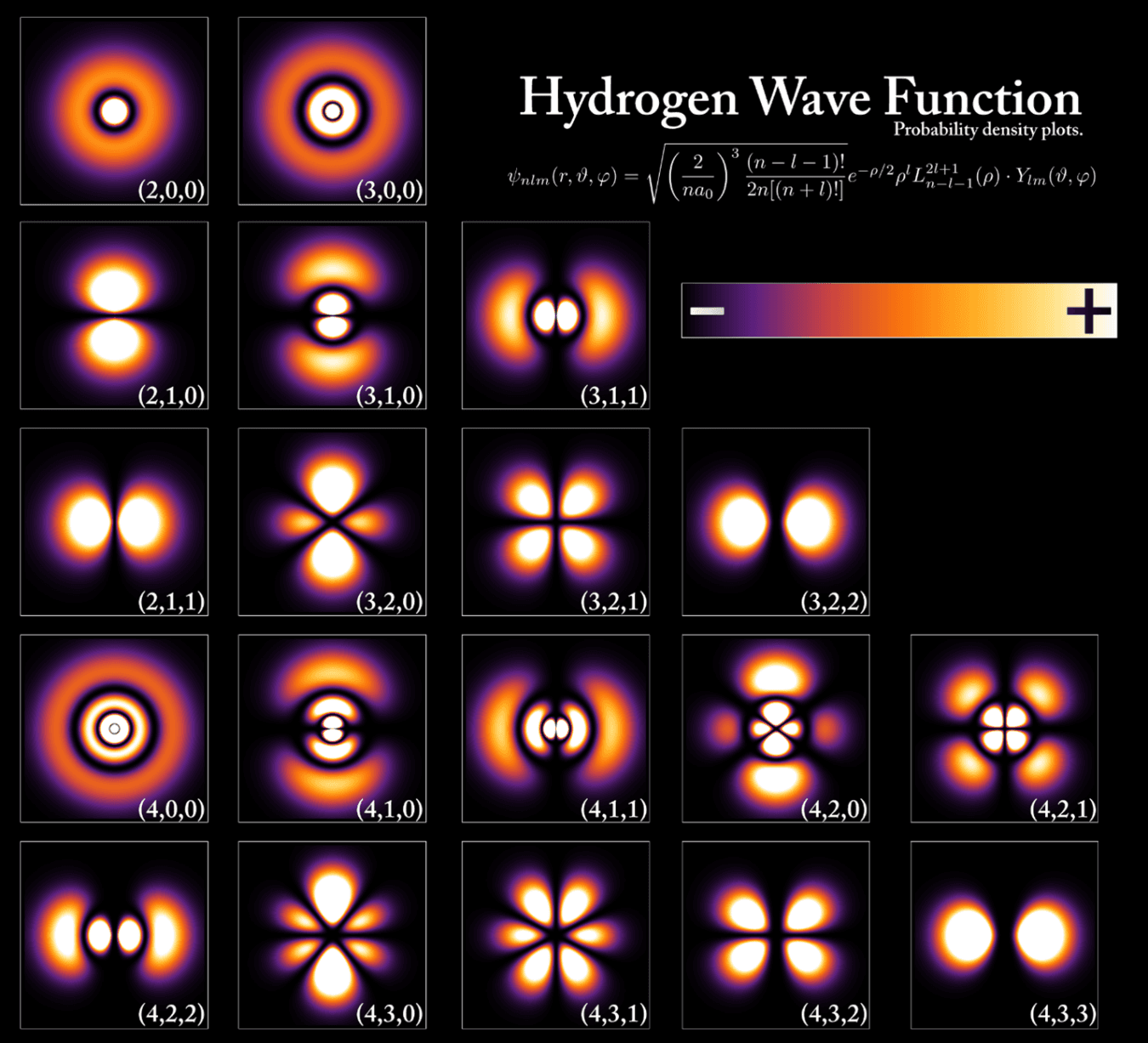
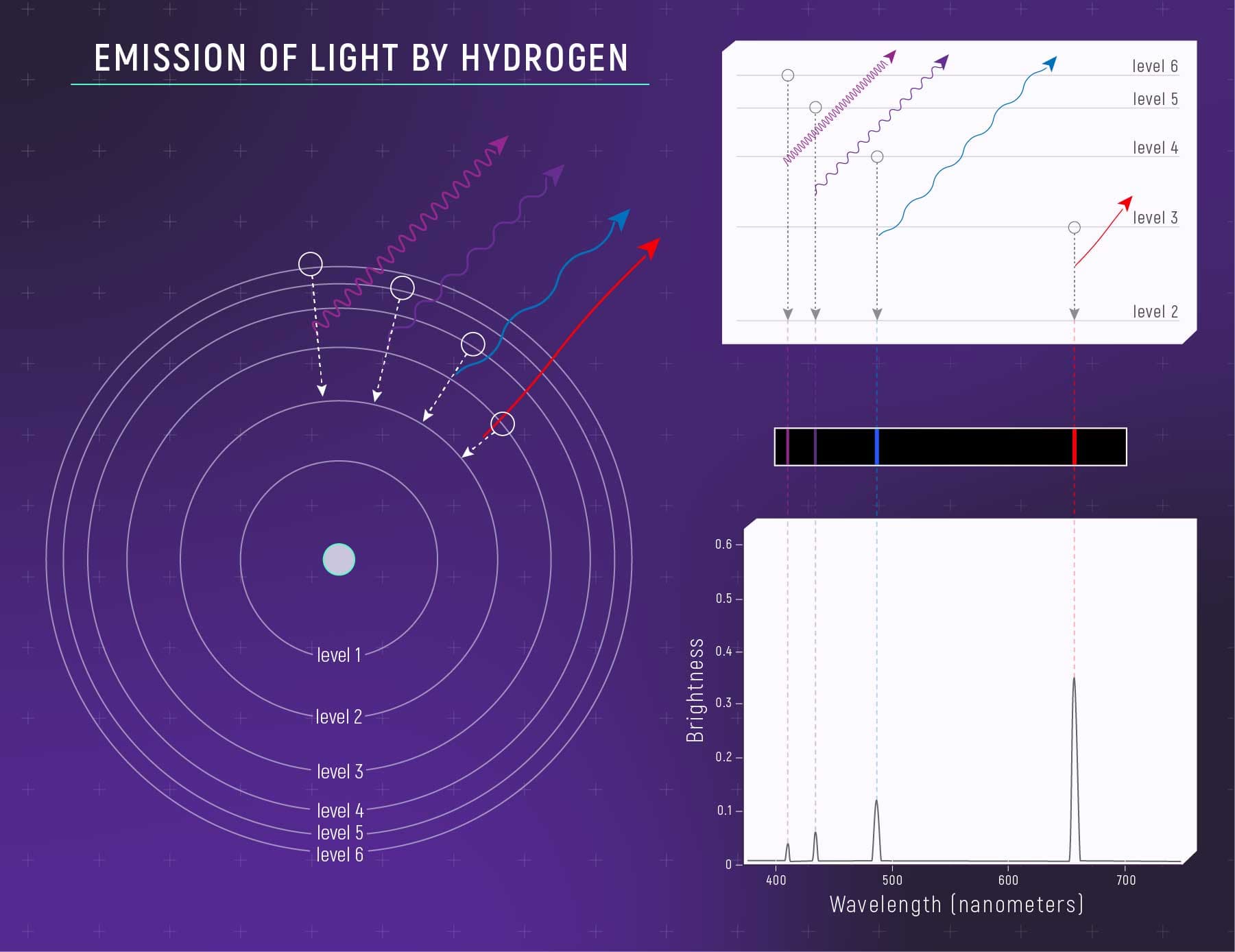
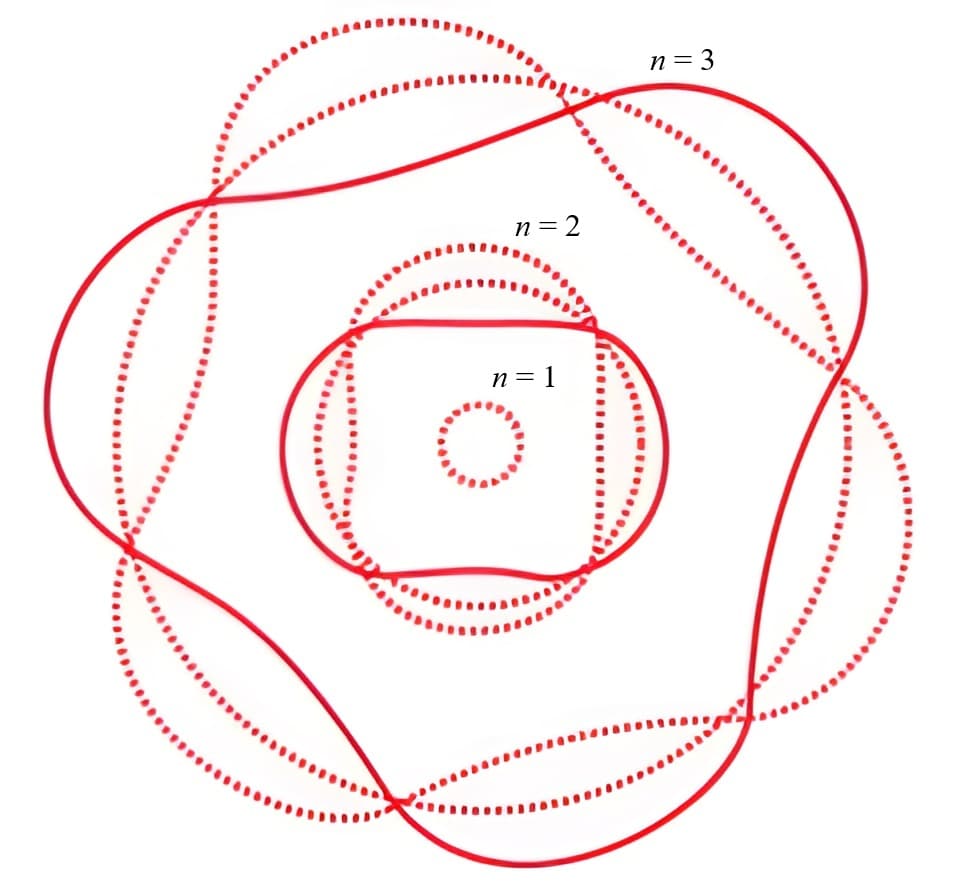
In quantum mechanics, matter is not in one location until it's measured
The probability that a particle is located at a given point is described by a wave function, in which peaks correspond to high probability and valleys to low probability. The wave nature of matter introduces uncertainty, allows it to exhibit complex behavior, such as interfering with itself, and also explains the observed structure of atoms.