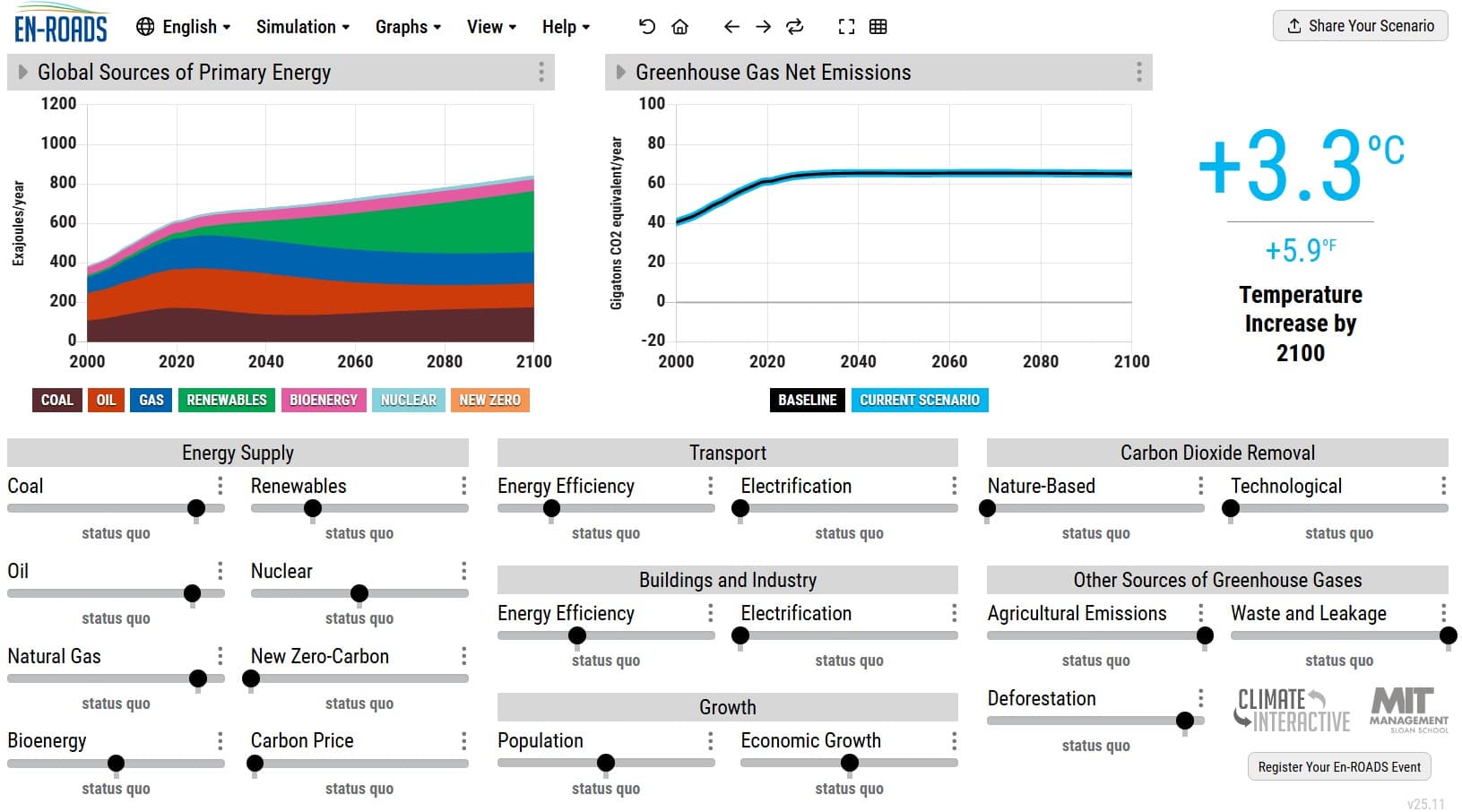
Glow sticks feed energy released in chemical reactions to fluorescent dyes
When bent, the glass tube inside the stick breaks, releasing hydrogen peroxide into the surrounding solution. The ensuing reaction creates unstable molecules that decompose and release energy, which is then absorbed by molecular dyes that emit light when their excited electrons return to lower energy states.